AI-Discovered Drug Shows Promise in Pulmonary Fibrosis Trial
Sonic Intelligence
An AI-generated drug, rentosertib, shows potential in treating idiopathic pulmonary fibrosis (IPF) in a phase 2a trial.
Explain Like I'm Five
"Scientists used a smart computer to find a new medicine for a bad lung disease. The medicine seems to help people breathe better, but they need to test it more to be sure it works."
Deep Intelligence Analysis
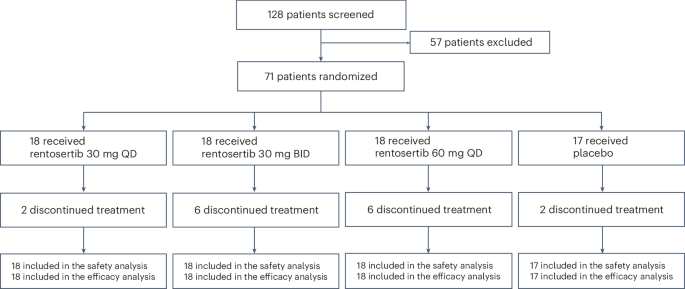
The trial results showed that rentosertib was safe and well-tolerated, with low rates of serious adverse events. Notably, patients receiving the highest dosage of rentosertib (60 mg QD) experienced an increase in forced vital capacity compared to the placebo group, suggesting a potential improvement in lung function. While these results are encouraging, it's important to note that this was a phase 2a trial, and further investigation in larger-scale clinical trials is necessary to confirm the drug's efficacy and safety.
The development of rentosertib highlights the potential of AI to accelerate and reduce the costs associated with drug discovery. By leveraging AI platforms, researchers can more rapidly identify and optimize therapeutic candidate compounds, potentially leading to the development of new treatments for a wide range of diseases. However, it's also important to acknowledge that AI-discovered drugs have experienced similar levels of phase 2 trial failure as non-AI-discovered drugs, underscoring the need for rigorous clinical testing and validation.
*Transparency Disclosure: This analysis was formulated by an AI assistant to provide an executive-level summary of the provided news article. The AI is trained to extract key facts and implications, and present them in a structured format. While efforts have been made to ensure accuracy, the AI-generated content should be reviewed by a human expert for complete factual correctness.*
Impact Assessment
This trial represents a significant step forward for AI in drug discovery, demonstrating the potential to accelerate the development of novel therapeutics. Rentosertib could offer a new treatment option for IPF, a debilitating lung disease with limited effective therapies.
Key Details
- Rentosertib is a first-in-class AI-generated small-molecule inhibitor of TNIK.
- The phase 2a trial tested rentosertib on patients with idiopathic pulmonary fibrosis (IPF).
- The trial showed increased forced vital capacity at the highest dosage (60 mg QD) compared to placebo.
- Treatment-related serious adverse event rates were low and comparable across treatment groups.
Optimistic Outlook
The positive results from the phase 2a trial warrant further investigation of rentosertib in larger-scale clinical trials. If successful, this AI-discovered drug could significantly improve the lives of patients with IPF and validate the role of AI in pharmaceutical research.
Pessimistic Outlook
Despite the promising results, further trials are needed to confirm the efficacy and safety of rentosertib. The drug's development could still face setbacks, and there is no guarantee that it will ultimately become a widely available treatment for IPF.
Get the next signal in your inbox.
One concise weekly briefing with direct source links, fast analysis, and no inbox clutter.
More reporting around this signal.
Related coverage selected to keep the thread going without dropping you into another card wall.